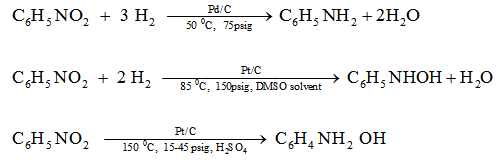
3. Hydrogenation of nitrobenzene
Hydrogenation of nitrobenezene shows how wide variety of products can be obtained by using different catalysts and reaction conditions.
Catalysts
Hydrogenation catalysts can be grouped in two categories as shown in Fig 1. Wide variation in commercial catalysts is available. Surface area, pore structure and presence of surface functional groups in supports greatly influence catalysts properties.
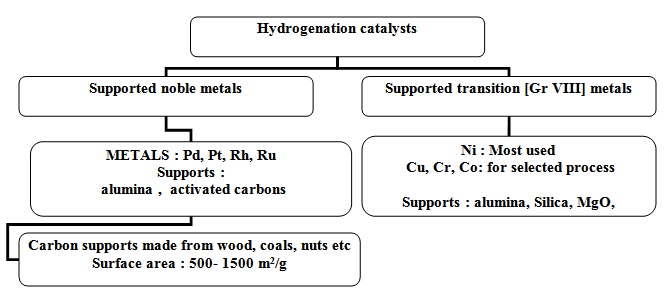
Fig. 1. Catalysts design for hydrogenation catalysts
Raney Ni
Raney Ni is a very important hydrogenation catalyst. Uniqueness of this catalyst is that it is a bulk unsupported catalyst with high surface area. The advantages include absence of support, minimum side reactions and more easy reduction of Ni in absence of interaction with supports. Raney Ni is typically prepared by the following steps:
- 1. Bulk Ni and Al metals in a 50-50 mixture are melted together.
2. Molten metals are poured into water producing fine grains
3. 20 % NaOH is added to leach Al from alloy leaving a porous matrix rich in Ni 90-95 % with a high surface area ~ 100 m2/g
4. Raney nickel is stored in an inert atmosphere such as water or fat oil to prevent reoxidation
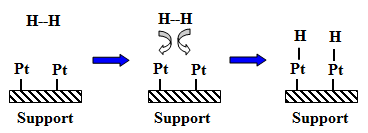
Fig. 2. Dissociative adsorption of hydrogen over Pt active site