Hydrogenation and dehydrogenation
Hydrogenation and dehydrogenation are the oldest catalytic processes. Catalytic hydrogenation reactions are used for production of fine chemicals, in pharmaceutical industry and polymer industry and for the production of edible and non edible fats and oils. Dehydrogenation reactions are used for production of light alkene C3-C4 for preparation of acrylonitrile, oxoalcohols or propylene oxides and production of C4-C8 for detergents. The process is also used for preparation of polypropylene, styrene, aldehydes, ketones etc.
Hydrogenation
Some of the hydrogenation processes are:
1. Hydrogenation of alkenes and alkadienes

Example
a. hydrogenation of ethene to ethane
- C2H4 + H2 = C2H6
b. hydrogenation of cyclohexene to cyclohexane
- C6H10 + H2 = C6H12
2. Hydrogenation of aromatics
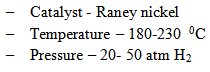
Example : Benzene to cyclohexane which is the precursor for nylon polymer.
- C6H6 + 3 H2 = C6H12