Dehydrogenation
Catalysts
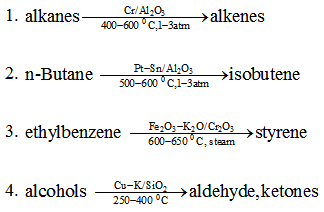
Important catalysts include chromic oxide/alumina and zirconia, Mo/alumina, Pt-Sn/alumina depending on the type of dehydrogenation reaction. Examples of important dehydrogenation reactions & catalysts are given below :
Kinetics and Mechanism
Dehydrogenation reaction can be modeled based on Langmuir-Hinshelwood kinetics. The Langmuir-Hinshelwood model of isobutane dehydrogenation proposed by Cortright et al. [2] for Pt-SnO2 based catalysts is given as
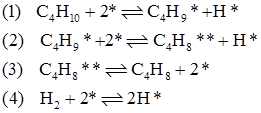
Where ‘*' is the active site. The second step of dissociative adsorption of isobutane is the slow and rate determining step. The corresponding rate expression is given as
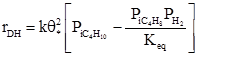
where ![]() is the net dehydrogenation rate, k is the rate constant for the dissociative adsorption of isobutane, Keq is the overall equilibrium constant for isobutene dehydrogenation, and
is the net dehydrogenation rate, k is the rate constant for the dissociative adsorption of isobutane, Keq is the overall equilibrium constant for isobutene dehydrogenation, and ![]() is the fraction of sites that is free of adsorbed species.
is the fraction of sites that is free of adsorbed species.
Deactivation
Main problem for dehydrogenation reactions is rapid deactivation by coke deposition requiring very frequent regeneration