Oxidation reaction
Oxidation processes consist of oxidation of inorganic compounds and partial oxidation of organic compounds. Oxidation of inorganic compounds is used to produce chemical feed stocks and is among the oldest and largest processes. Production of sulphuric acid by SO2 oxidation and nitric acid by ammonia oxidation are amongst the major basic chemical industry. Oxidation processes will be illustrated by few examples.
Oxidation of inorganics
SO3 production
SO2 is oxidized to SO3 which is further converted to sulphuric acid. This process is one of the highest tonnage processes in the world.
SO2 + ½ O2 = SO3
Catalyst for this process is vanadium potassium sulfate supported on silica rich carrier such as natural clay. Minimum inlet temperature required for vanadium based catalysts is 400°C. For single pass operation at 400°C the temperature rise to 600°C and the conversion is limited to 60% by equilibrium. So multiple pass reactors with intermittent heat exchanger is used to achieve near 100 % conversion.
Mars and Maessen [4] proposed a two step redox mechanism as shown below. Second step is the rate determining.
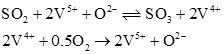
Using Langmuir-Hinshelwood model, rate is given as
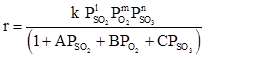
Where l and m lies between 0.5 to 1 and n lies between -1 to 0.