Partial oxidation of organics
Methanol to formaldehyde
Methanol is partially oxidized to produce formaldehyde. Formaldehyde is used for production of urea, disinfectant, preservative etc.
CH3OH + 0.5 O2 = HCHO + H2O
The process is carried out at 600-7000C temperature at near atmospheric pressure using silver needles or gauges catalysts. As a result of high process temperature, the unsupported silver catalyst tends to sinter resulting in increased pressure drop and reduced activity. To minimize these adverse effects without compromising adequate contact of reactants with the catalyst, a shallow (4 to 5 cm thickness) but wide bed (~ 3m) of Ag needles or gauge is used. The unsupported silver catalyst is sensitive to S, N, halide compounds. Methanol oxidation process at dilute condition is carried out over Fe2O3 -Co2O3 -MoO3 catalyst at lower temperature of 300 – 4000C at atm pressure.
Ethylene to ethylene oxide
Ethylene oxide is used to produce ethylene glycol. Polyethylene glycols are important monomers for liquid plasticizers, lubricants. Ethylene oxide is produced by partial oxidation of ethylene and is a highly exothermic process. Process is carried out at 200 – 2500C temperature over silver supported on alumina catalysts. The process is always associated with secondary oxidations of ethylene oxide (equation 6) or total oxidation of feed to carbon oxides and water (equation 7) as shown below. This can be minimized by controlling process conditions and proper catalyst selection.
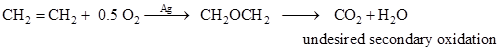 |
(6) |
| (7) |