Kinetics and mechanism
During hydrogenation of alkene over noble metal based catalysts, at first hydrogen get adsorbed dissociatively on the metal sites (Fig. 2). Dumesic et al. [1] reported that adsorption of hydrogen follows different kinetics at low alkene coverage and at high coverage. For hydrogenation of ethylene two types of site for hydrogen adsorption was proposed. The proposed mechanism is shown below. The two types of site for hydrogen adsorption are represented by ‘S' and ‘*'.The site for adsorption of hydrogen on a surface partially covered with ethene was represented as ‘S', whereas hydrogen adsorption site on a clean surface was designated as ‘*'. At adsorption sites, ‘*', both the hydrogen and ethene can get adsorbed but at ‘S' site only hydrogen can adsorb .

Hydrogen adsorbed dissociatively on the Pt surface at ‘S' or ‘*' adsorption sites forming H-S' or ‘H*'. The adsorbed hydrogen atom ‘H-S' or ‘H*' were further assumed to be activated on another site Sˈas shown by step 2 or 7. The activated hydrogen H-Sˈ then can react with C2H4 ** to give C2H5** (step 4) and then with C2H5** to give C2H6 (step 5). The Step 5 was considered as slowest and rate determining step.
Hydrogenation of organic compounds is usually conducted in liquid phase in a slurry reactor. Most hydrogenation reactions are highly exothermic and hence careful temperature control is required. Solvent is used to affect the product selectivity and to absorb the liberated reaction heat. Hydrogenation reactions in the slurry reactor are typically limited by mass transfer of H2 from gas bubbles through liquid to each suspended catalyst particle. For hydrogenation reaction to occur, the hydrogen has to be transported from bulk phase to liquid phase through gas – liquid interface and then from bulk liquid to surface of solid catalyst for final reaction. The Fig. 3 schematically shows the various concentrations of hydrogen at different positions for a single bubble and particle combination. The Cg, CL and Cs shown in the figure represent concentration of hydrogen in gas bubble, liquid and catalyst surface respectively.
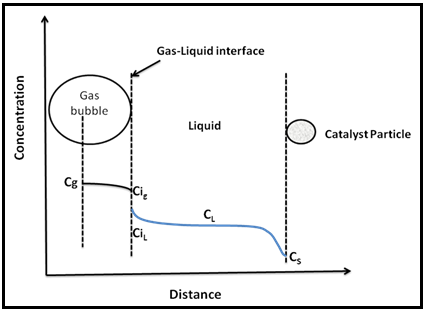
Fig. 3. Concentration profile of gaseous reactant in slurry reactor
For first order irreversible catalytic reaction, the surface reaction rate per unit volume of bubble free slurry may be written as
| (1) |
Where,
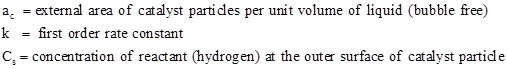
The rates of three mass transfer processes for hydrogen may be expressed as
| for transport from bulk gas to gas – liquid interface | (2) |
| for transport from gas – liquid interface to bulk liquid | (3) |
| for transport from bulk liquid to catalyst surface | (4) |
where, ag is the gas bubble – liquid interface area per unit volume of bubble free liquid and kg, kL and kc are the appropriate mass transfer coefficient.
H2 pressure decreases across the gas film due to film diffusional resistance. At the gas liquid interface, a concentration of hydrogen in the liquid is determined in accordance with Henrys law. If equilibrium exists at the bubble-liquid interface Cig and CiL are related as ![]() (5)
(5)
Where, H is known as Henry´s constant. From equations 1,2,3,4 and 5 the Cig , CiL , CL and Cs can be eliminated and the rate can be expressed in terms of reactant in gas as
![]()
Where ko is given as
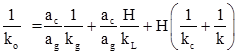
The ko is the function of three mass transfer coefficients, the reaction rate constant and area ratio of catalyst and gas bubble in contact with liquid. The values of mass transfer coefficients can be obtained from several correlations available in literature.
Deactivation
Deactivation of catalysts mainly occurs by poisoning of metal surface with sulfur, halogens, phosphorus and nitrogen compounds. As the hydrogenation reactions occur in slurry condition, mechanical attrition is another major source of loss of catalysts. In addition, pore blockage may occur due to coke deposition.