Orbitrap
Orbitrap is an electrostatic ion trap. It has a barrel containing a spindle-shaped electrode at the centre. The spindle electrode is held at a constant negative voltage (-3200 V) for positive ion mode MS. Ions enter the orbitrap tangentially and get trapped by revolving around the spindle shaped electrode (Figure 12.7). The ions can be ejected out by applying the radiofrequencies of suitable frequency to the central electrode.
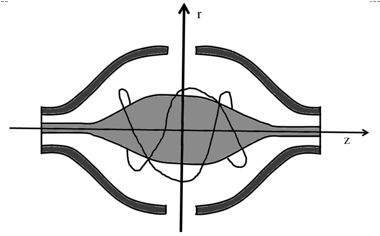
Figure 12.7 A schematic diagram of an orbitrap |
Mass spectrometry coupled with chromatography
Mass spectrometers have been successfully coupled with the liquid and gas chromatographic methods. Chromatographic methods separate the compounds based on the differences in their physico-chemical properties. A complex mixture of compounds can be resolved into pure components using one or more chromatographic methods. Though excellent in separating the molecules, chromatographic methods do not allow identification of the unknown compounds in the mixture. The separated components can be collected and identified using MS. It is also possible to couple the mass spectrometers with the chromatographic methods. Gas chromatography (GC) and liquid chromatography (LC) coupled with mass spectrometers have emerged as very powerful analytical tools. GC allows easy interfacing with the mass spectrometers; a gas chromatographic column can directly be coupled to the ionization source of the MS. Interfacing the LC with MS, however, is not as straightforward. We shall not be discussing the different types of interfaces of LC and MS. As it allows studying the large, polar, non-volatile, and thermolabile compounds, LC-MS is more widely used compared with GC-MS. Electrospray ionization is one of the softest ionization methods and probably the most widely used ionization method for LC-MS. LC-MS and GC-MS therefore allow determining the molecular masses of the eluants in real-time. The ions separated by MS can be fragmented in a collision cell; identification of these fragments by another mass analyzer allows identification of the compounds (tandem mass spectrometry). We shall see in the next lecture how fragmented ions help in the identification of the compounds.