Consider the rods in x-z plane to be at a positive potential, U and the rods in y-z plane to be at a negative potential –U. An a.c. potential is subsequently applied to the rods such that the a.c. potential on the rods in x-z plane is 180° out of phase than that applied on the rods in the y-z plane. The potential on the rods in x-z and y-z plane can therefore be represented as
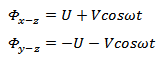
The values of U range from 500 – 2000 volts while V ranges from 0 – 3000 V. The gas phase ions are accelerated and introduced into the quadrupole as a focused beam. To understand how the ions are separated inside a quadrupole, let us consider the rods in the x-z plane and those in the y-z planes separately. The rods in x-z plane have a positive d.c. potential and an a.c. potential that will periodically make the overall potential negative (Figure 12.5B). The ions will respond to the changes in the potential. If a cation is very heavy or the frequency of the a.c. potential is very high, the cation remains largely unaffected and experiences only the average potential. This causes the ion to get focused towards the centre. If a cation is very light, it will readily respond to the changes in the potential and can accelerate towards the rods during negative potential and collide with them. Collision of the cation with the quadrupole rods during negative potential depends on the magnitude of the potential on the rods, frequency of the a.c. potential, mass of the cation, charge on the cation, and the position of the cation in the quadrupole. Let us now turn our attention towards the rods in the y-z plane. These rods have a negative average potential; the heavier cations will therefore get accelerated towards the rods and collide with them. Lighter cations will respond to the a.c. potential and get focused towards the centre. We can say that the x-z rods filter out the lower masses while y-z rods filter out the higher masses. The four rods together can therefore be used to allow the passage of a very small range of masses. A quadrupole therefore acts as a mass filter.
Ion trap
As the name suggests, ion trap mass analyzers trap the ions inside them. An ion trap can either be a 2D or a 3D ion trap. A 3D ion trap is basically a quadrupole in 3 dimensions. It has a circular electrode (also called a ring electrode) with two ellipsoid electrodes as its caps (Figure 12.6).
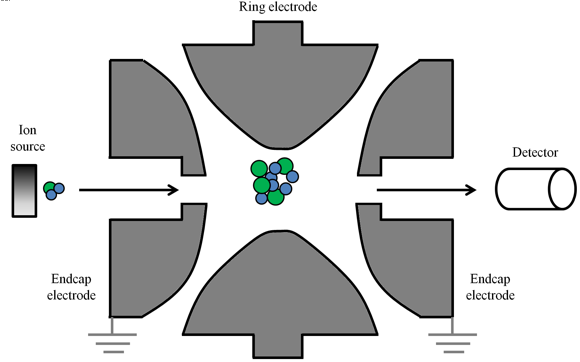
Figure 12.6 A diagrammatic representation of an ion trap
All the three electrodes have hyperbolic surfaces. Unlike linear quadrupole (quadrupole with four parallel rods) wherein electrostatic forces act in two axes, forces act in all the three axes in an ion trap. This implies that the stable trajectories of the ions cause them to be trapped. Ion traps provide higher sensitivity and are useful in tandem mass spectrometry; ions of desired mass can selectively be allowed to escape the trap by varying the ac potential; the escaped ions can then be analyzed by another mass analyzer attached in tandem.