
Fig 3-4.2.1: Set up for Electro-blotting system
5. For using alkaline transfer methods, the DNA gel is placed into an alkaline solution (like that of sodium hydroxide) causing denaturation of the double-stranded DNA. Denaturation in an alkaline environment enhances the binding between the negatively charged DNA and the positively charged membrane, causing separation to single DNA strands for further hybridization to the probe, alongside destroying any residual RNA that may persist in DNA. The membrane is washed with buffer to remove unbound DNA fragments.
6. The membrane which contains the transferred fragments is heated in presence or absence of vacuum at 80°C for 2 hours or exposed to ultraviolet radiation (nylon membrane) for permanent attachment of the transferred DNA to the membrane.
7. The obtained membrane is then hybridized with a probe (a DNA fragment with a specific sequence whose presence in the target DNA is to be determined).
8. Labeling of the probe DNA is done for easy detection, usually radioactivity is incorporated or the molecule is tagged with a fluorescent or chromogenic dye. The hybridization probe may be made of RNA, instead of DNA in some cases where the target is RNA specific.
9. Washing of the excess probe from the membrane is done by using saline sodium citrate(SSC buffer) after the hybridization step and the hybridization pattern is studied on an X-ray film by autoradiography (for a radioactive or fluorescent probe), or via color development on membrane if a chromogenic detection method is employed.
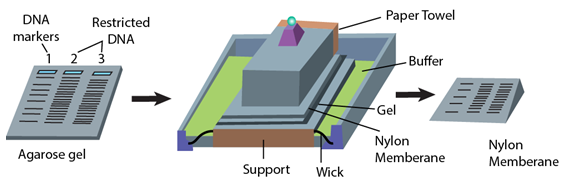
...................................Fig 3-4.2.1: Steps of Southern Hybridization