2. Gibberlin:
Gibberellins (GAs) are a group of diterpenoid acids that function as plant growth regulators inflencing a range of developmental processes in higher plants including stem elongation, germination, dormancy, flowering, sex expression, enzyme induction and leaf and fruit senescence.
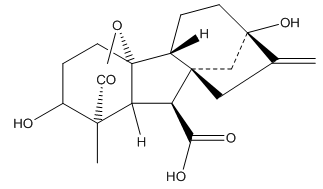
Figure 5: Gibberellin
Discovery of Gibberellin: Kurusawa, a Japanese plant pathologist, discovered gibberellin in 1926. When he was working in the rice fields, he observed that some of the rice seedlings grew much taller than the others which were found to be infected by a fungus, Gibberella fujikuroi. Yabuta and Yabuta and Sumiki (1938) demonstrated that some substances secreted by the fungus were probably responsible for more elongation (growth) of the seedlings. Till date, about 62 different gibberellins are known out of which 25 have been isolated from the fungus Gibberella fujikuroi.
Chemical structure of Gibberellins:
All gibberellins are derived from the ent-gibberellane skeleton. During the synthesis of gibberellins, the central 6-member ring is reduced to 5 carbons to make the basic gibberellin. The carbons are numbered 1 through 20. Gibberellins are diterpenes synthesized from acetyl CoA via the mevalonic acid pathway. They all have either 19 or 20 carbon units grouped into either four or five ring systems. The fifth ring is a lactone ring. They have been depicted in Figure 7.
Biosynthesis: In the formation of gibberellins 3 acetyl CoA molecules are oxidized by 2 NADPH molecules to produce 3 CoA molecules as a side product and mevalonic acid. Further Mevalonic acid is then phosphorylated by ATP and decarboxylated to form isopentyl pyrophosphate. 4 of these molecules form geranylgeranyl pyrophosphate which serves as the donor for all GA carbon atoms. This compound is then converted to copalylpyrophosphate which has 2 ring systems. Copalylpyrophosphate is then converted to kaurene which has 4 ring systems.
Subsequent oxidations reveal kaurenol (alcohol form), kaurenal (aldehyde form), and kaurenoic acid respectively. Kaurenoic acid is converted to the aldehyde form of GA12 by decarboxylation. GA12 is the 1st true gibberellane ring system with 20 carbons. From the aldehyde form of GA12 arise both 20 and 19 carbon gibberellins but there are many mechanisms by which these other compounds arise. Transport of Gibberellin in plants is non-polar. It moves from one part to another in the phloem. Due to the lateral movements between the two vascular bundles, gibberellins are translocated in the xylem.