| which we have already seen in section 3.3. |
|
A nice way of visualization of these for l = 1 is given in fig 3.6. This is commonly referred to as space quantization. For l = 0 , the angular momentum = 0 . For l = 1 and ml = 0, the angular momentum is oriented along a radius vector on the sphere such that its projection on the z axis is zero. For l =1 and ml = 1, the angular momentum vector is in the upper cone such that the projection on z axis = 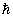 . For l = 1 and ml = -1, the angular momentum is in the lower cone with a projection on z axis = - . For l = 1 and ml = -1, the angular momentum is in the lower cone with a projection on z axis = - 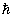 .This implies that all regions of space are not accessible to the .This implies that all regions of space are not accessible to the 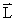 vector . Only some regions are accessible and this is turned space quantization vector . Only some regions are accessible and this is turned space quantization |