| Problems |
| 3.1) |
An electron is moving in a circular orbit of radius 2 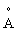 with a speed of 10 6 m/s.
What is its angular momentum and momentum of inertia? (m e = 9.1 x 10 -31 kg) with a speed of 10 6 m/s.
What is its angular momentum and momentum of inertia? (m e = 9.1 x 10 -31 kg) |
| |
|
| 3.2) |
What is the operator for the x component of angular momentum L x ? |
| |
|
| 3.3) |
If l = 3, what are the admissible values of m l ? For each m l what is the eigenfunction 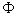 ( ( 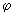 )? )? |
| |
|
| 3.4)
|
Verify that the functions in Table 3.1 for l = 1 and m l = 0 and l = 1 and m l = 1 satisfy the equation (3.15.) |
| |
|
| 3.5) |
For the electron in problem 1, what are the quantized values of angular momentum and energy for l = 0, 1 and 2? |
| |
|
| 3.6) |
The region of space where the wavefunction is zero is called a node. For the function in Table 3.1 for l = 0 and m l = 0, what is the shape of the node? |
| |
|
| 3.7) |
Real functions can be obtained by combining the functions for positive and negative values of ml using........ ei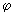 = cos = cos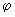 + sin + sin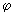 ; cos ; cos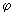 = ½ (ei = ½ (ei 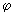 + e -i + e -i 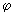 ); and sin ); and sin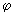 = (ei = (ei 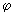 - e -i - e -i 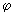 ) /(2 i). Obtain two real solutions for l = 1 by suitable linear combinations of the two functions for m l = 1 and m l = -1. Repeat the process for l = 2 and m l = ) /(2 i). Obtain two real solutions for l = 1 by suitable linear combinations of the two functions for m l = 1 and m l = -1. Repeat the process for l = 2 and m l = 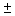 2. 2. |
| |
|
| 3.8) |
Using the function (3/4
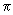 ) 1/2 cos ) 1/2 cos 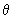 , estimate the probability of finding the electron in an angle interval of d , estimate the probability of finding the electron in an angle interval of d 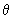 = 0.2o when = 0.2o when 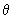 = 0o , = 0o , 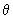 = 45 o and = 45 o and 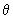 =90 o. Repeat the calculation for the function for l = 2, m l = 0 for d =90 o. Repeat the calculation for the function for l = 2, m l = 0 for d 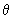 = 0.2o and = 0.2o and 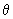 = 0, 22.5o ,45 o ,67.5 o and 90 o .Use the formula of Eq (3.25) without the d = 0, 22.5o ,45 o ,67.5 o and 90 o .Use the formula of Eq (3.25) without the d 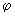 part. part. |
| |
|