| It is seen that larger the values of A, J0 and 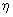 , faster the corrosion rate. Even if the emf is smaller, the process with larger J0 will occur more rapidly. E.g., the J0 for reaction (a) in the figure is 10 -6 Acm-2 and for (b) it is 1014 A cm -2 and kinetically, the hydrogen evolution dominates the corrosion process. , faster the corrosion rate. Even if the emf is smaller, the process with larger J0 will occur more rapidly. E.g., the J0 for reaction (a) in the figure is 10 -6 Acm-2 and for (b) it is 1014 A cm -2 and kinetically, the hydrogen evolution dominates the corrosion process. |