| The Nernst Equation |
| In the previous chapter, we have seen that for a reaction such as |
| |
|
aA + bB 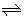 cC +dD, cC +dD, |
(22.4) |
| which is at any stage of completion, the free energy change is given by |
| |
| 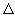 G = G = 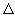 G o + RT ln [C] c [D] d / [A] a [B] b G o + RT ln [C] c [D] d / [A] a [B] b
|
(22.5) |
where the bracketed quantities [A], [B], [C] and [D] refer to the actual activities (or concentrations in ideal solutions, pressures or fugacities (divided by p o ) for gases) of these species in the reaction mixture. The quotient on the right hand side is denoted by Q and if the reaction has reached equilibrium, it becomes equal to K, the equilibrium constant. Using the relation between the change in free energy and emf, i.e., 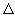 G = -nF and G = -nF and 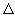 G o = -nFo, G o = -nFo, |
| we have |
| |
| E = E o (RT/nF) lnQ |
(22.6) |
And at equilibrium, 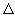 G = 0, E = 0 and G = 0, E = 0 and |
| E o = (RT / nF ) lnK |
(22.7) |
| |
|
| Or K = exp [ n F E o / RT ] |
(22.8) |
| |
The equation (22.6) / (22.7) is referred to as the Nernst equation. This is the fundamental equation of equilibrium electrochemistry. Using this equation, the cell potential at any stage of the reaction can be calculated. At 298K and 1 atm (101.3 kPa ) pressure, RT / F = 8.314 J K -1 mol -1 . 298 K / (96500 C/mol) = 0.02569 V and the Nernst equation becomes |
| |
| E = E o (0.02569 / n) ln Q or |
(22.9) |
| |
|
| E = E o (0.05916 /n ) log 10 Q |
(22.10) |
| The above equations are convenient forms of the Nernst equation for calculations done at a room temperature of 25o C. |
| |