| Objectives |
| After studying
this Lecture you will be able to |
 |
Distinguish between electrolytic cells and galvanic cells. |
 |
Write the cell representation for electrochemical reactions. |
 |
Derive the Nernst equation. |
 |
Use the Nernst equation for calculating the electromotive force (emf) of a cell for any combination of activities of |
| |
solution species. |
 |
Evaluate 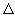 rG, rG, 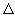 rH and rH and 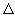 rS form dE /dT. rS form dE /dT. |
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|