| Cell Representation and sign convention |
| The above Daniell cell may be conveniently represented in a compact form as |
| Zn(s)| Zn2+ (C1 ) || Cu2+ (C2 ) | Cu(s) |
| |
| Here, the vertical line denotes a boundary separating two phases. The double vertical lines indicate a salt bridge. On either side of the salt bridge are two half cells, or electrodes. On the left hand side (L), oxidation (O) occurs. This electrode is referred to as the anode (A) and it is the negative (N) terminal. A simple way to remember this arrangement is to remember the word LOAN (left, oxidation, anode, negative). Automatically this implies that at the other electrode on the right (R), reduction (R) occurs; the electrode is called the cathode (C) and it is the positive terminal(P), (RRCP). |
The reactions at the anode and the cathode are respectively, |
| Zn(s) = Zn2+ + 2e |
(22.2a) |
| |
| Cu2+ + 2e = Cu(s) |
(22.2b) |
| |
The emf (electromotive force) of this cell is 1.103V. The potential (emf) of a single electrode or a half cell can not be measured; we always need to electrodes to obtain an emf. |
| |
Instead of listing the potentials of all pairs of cells, it is far more convenient to take any one half cell as the standard and list all the other electrode potentials (actually reduction potentials, as recommended by the international Union of Pure and Applied Chemists, IUPAC) with reference to this standard. This reference electrode is taken as the standard hydrogen electrode (SHE) whose emf is assigned a reference value of 0 V . |
| |
H + + e 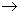 1 / 2 H2 (g), E o H + / H2 = 0 V 1 / 2 H2 (g), E o H + / H2 = 0 V |
(22.3) |
| |
|