The last section dealt with calculating 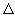 rS and rS and 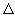 rH for reactions, if in addition to E (which gives rH for reactions, if in addition to E (which gives 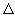 rG), the temperature dependence of E, dE /dT can be measured experimentally. This option provides a very accurate method for determining rG), the temperature dependence of E, dE /dT can be measured experimentally. This option provides a very accurate method for determining 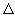 rG, rG, 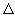 rH and rH and 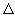 rS for electrochemical reactions. rS for electrochemical reactions. |