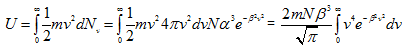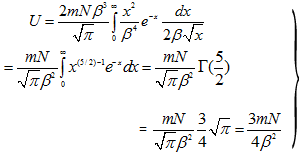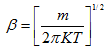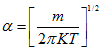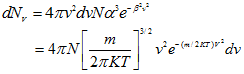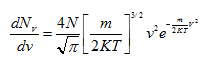from which
|
(4.98) |
The translational K.E. of the molecules of a gas is given by
|
(4.99) |
Let x = β2v2, as before
|
|
(4.100) |
Now,
|
(4.101) |
Hence,
|
(4.102) |
From Eq.(4.98)
|
(4.103) |
Substituting these values in Eq.(4.93),
|
(4.104) |
or,
|
(4.105) |
This equation is known as Maxwell Boltzmann velocity distribution function.