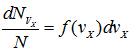It was shown that the rms velocity of the molecules of gas is related to the gas temperature. If all the molecules of a gas at a certain temperature move at the same speed, then the rms value indicates the magnitude of the velocity of all the molecules. However, the speeds of gas molecules vary widely, and it is thus necessary to determine the velocity distribution of the molecule, so that the number of molecules moving with any particular velocity can be determined.
Let us consider a volume of gas at a constant temperature, the molecules of which are moving at different velocities. The instantaneous velocity vector of each molecule is revolved into components vx, vy and vz. Let us imagine a velocity space (Fig.6.7) so that the space area of a sphere represents, at an instant, all molecules of equal velocities.
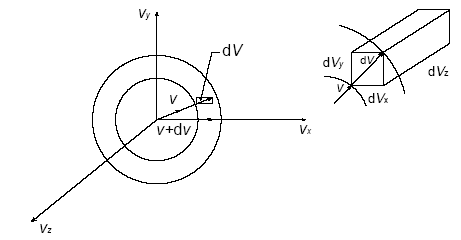
Fig. 4.12 Velocity Space
Each molecule has a representative point in velocity space. The number of molecules whose velocities lie between v and v + dv would be represented by the spherical strip of thickness dv, and be denoted by dNv. Since the total number of molecules N is very large, the strip dv, although small, still contains a large number of molecules. Let dNvx represent the number of molecules whose x -component velocities lie between vx and vx + dvx. Then the fraction ![]() is a function of the magnitude of vx and the distance dvx,
is a function of the magnitude of vx and the distance dvx,
or,
|
(4.66) |