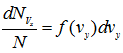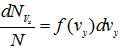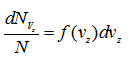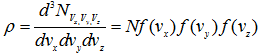where f(vx) is called the distribution function for x -component of velocity.
Similarly,
|
(4.67) |
and
|
(4.68) |
|
(4.69) |
Some of the molecules of ![]() have y -component velocities lying between vy and vy + dvy and let this be represented by
have y -component velocities lying between vy and vy + dvy and let this be represented by . A differential of second order
has been used, since this is a small fraction of an already small fraction, but still large enough to contain many molecules. Since the number of molecules is large, the following relation holds good.
(4.70) |
|
(4.71) |
Similarly, represent the number of molecules whose velocity components would lie between vx and vx + dvx, vy and vy + dvy and vz and vz + dvz. Therefore,
|
(4.72) |
The number of representative points per unit volume, or the density of point in velocity space, represented by ρ, is
|
(4.73) |
Since the velocity distribution is isotropic, the density is the same in any volume element so that
|
(4.74) |