In order to determine the structure of an organic compound, it is advised to proceed in the following manner.
-
The history of the compound has to be determined. This includes the source from which the compound is obtained, or in case of a synthetic compound the series of steps which were carried out to obtain the compound.
-
The UV-Visible spectrum may be recorded and λmax and its corresponding εmax is to be determined. It may give information about the degree of conjugation in the molecule and well as information about certain functional groups like carbonyl group.
-
The IR spectrum also provides information about functional groups such as carbonyl group (aldehydes, ketones, carboxylic acid and its derivatives) and amine functionality.
-
NMR spectrum provides the most exhaustive source of information regarding the structure of the molecule
As an example, if a C9H12O compound has strong infrared absorption at 3300 to 3400 cm-1 . The 13C NMR spectrum of this compound has six discrete signals. It's 1H NMR spectrum has three sets of lines: singlets at δ1.1 (6H), 1.9 (1H) and 7.3 (5H) ppm.
In this case the molecular formula has been deduced by mass spectroscopy. A calculation to determine the degree of unsaturation is usually helpful. It is defined as follows.
In this case the value comes out to be 4. This is indicative of a benzene ring or 4 double bonds or two triple bonds or a triple bond and a double bond. A strong absorption at 3300-3400 cm-1 indicates a O-H group. In the 1H spectrum, the five 5 peaks at 7.3 ppm are indicative of the presence of monosubstituted benzene ring. Similarly, a singlet at 1.1ppm is indicative of two methyl groups which are not connected to each other. The peak at 1.9 could be due to the O-H proton. Hence the structure that could be deduced is-2-phenylpropan-2-ol.
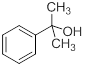 |
Now in the 13C NMR spectrum, this compound should show 6 signals which is same as observed. Hence the deduction is correct.
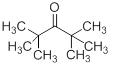
Similarly, if a compound C9H18O has a strong infrared absorption at 1710 cm-1 . Its 1H NMR spectrum has a single sharp peak (a singlet) at δ 1.2 ppm while 13C NMR spectrum shows three lines at δ210, 45 and 25 ppm. The logic for the elucidation of structure may be as follows:
Degree of unsaturation is 1. This surely means there is only one double bond in the molecule. A strong IR absorption at 1710 cm-1 indicates a carbonyl group. A peak in 1H spectrum at 1.2 ppm indicates the presence of a methyl group, however there must be multiple methyl groups with a symmetrical environment to satisfy the molecular formula. A further clue may be obtained from the 13C spectrum that there are two peaks in the aliphatic region (45 and 25 ppm) but only one peak in 1H spectrum. This means the other carbon (at 45 ppm) is a tertiary carbon. This overall scenario leads to the assumption of the presence of two t-butyl groups. Hence the compound is 2,2,4,4-tetramethylpentan-3-one.
 |
Text Books
• P. Y. Bruce and K J R. Prasad, Essential Organic Chemistry , Pearson Education, New Delhi, 2008.
• C. M. Loudon , Organic Chemistry , Oxford University Press, 2002, New Delhi.
• R. R. Morrison, R. N. Boyd and S. K. Bhattacharjee, Organic Chemistry , Dorling Kindersley (India) Pvt. Ltd, 2011, New Delhi.