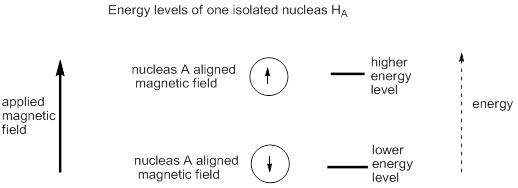
The second explanation takes into account the energy levels of the nucleus. Electronic energy levels on neighbouring atoms interact with each other and splitting to produce new molecular energy levels, some higher in energy and some lower in energy than the original atomic energy levels. When hydrogen nuclei are near each other in a molecule, the nuclear energy levels also interact and split and produce new energy levels. If a single hydrogen nucleus interacts with a magnetic field there are two energy levels as the nucleus can be aligned with or against the applied magnetic field, there is one energy jump possible, and there is a resonance at one frequency.
• This representation shows H
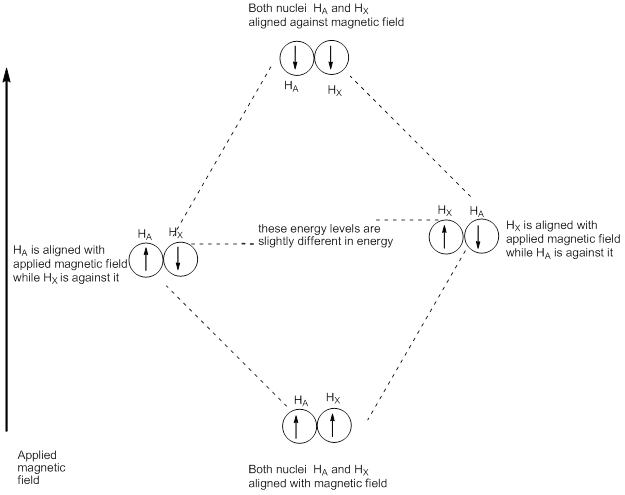
A protons independent of the influence of H
X. Each had two energy levels, each gave a singlet, and there were two lines in the spectrum. But, if each proton has hydrogen nucleus nearby and then there are now four energy levels. Each nucleus H
A and H
X can be aligned with or against the applied field. There is one most stable energy level where they are both aligned with the field and one least stable level where they are both aligned against. In between there are two different energy levels in which one nucleus is aligned with the field and one against. Exciting H
A from alignment with to alignment against the applied field can be done in two slightly different ways, shown as A
1 and A
2 on the diagram given below. The result is two resonances very close together in the spectrum. If there are two slightly different energy jumps to excite H
A, there must also be two slightly different energy jumps to excite H
X. The difference between A
1 and A
2 is exactly the same as the difference between X
1 and X
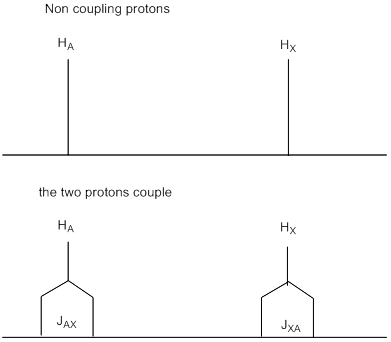
2. Each proton now gives two lines (a doublet) in the NMR spectrum and the splitting of the two doublets is exactly the same. This situation is described as coupling. We say ‘A and X are coupled' or ‘X is coupled to A'(and vice versa, of course). Now look back at the spectrum of cytosine at the beginning of this section. Each is split by the same amount and the separation of the lines is the coupling constant and is denoted by J. Coupling constants J are measured in Hz because the same number regardless of the frequency of the spectrometer.