9.5 Nuclear Magnetic Resonance Spectroscopy (NMR)
Nuclear magnetic resonance (NMR) allows us to detect atomic nuclei and say what sort of environment they are in, within their molecule. Thus, the hydrogen of, say, propanol's hydroxyl group is different from the hydrogens of its carbon skeleton. NMR (actually 1H, or proton, NMR) can easily distinguish between these two sorts of hydrogens. Moreover, it can also distinguish between all the other different sorts of hydrogen atoms present. Likewise, carbon (or rather 13C) NMR can easily distinguish between the three different carbon atoms.
-
Some atomic nuclei act like tiny compass needles and have different energy levels when placed in a magnetic field. Nuclear energy levels are quantized, just like the energy levels of an electron, and there are only certain specific energy levels it can adopt. Some nuclei (including ‘normal' carbon-12) do not interact with a magnetic field at all and cannot be observed in an NMR machine. 1H and 13C do interact and have just two different energy levels. When a magnetic field is applied to these nuclei, they can either align themselves with it, which would be the lowest energy state, or they can align themselves against the field, which is higher in energy. nucleus in a magnetic field, the difference in energy between the nuclear spin aligned with and against the applied field depends on how strong the magnetic field is, and also on the properties of the nucleus itself. The stronger the magnetic field we put our nucleus in, the greater the energy difference between the two alignments. However, the energy difference between the nuclear spin being aligned with the magnetic field and against it is really very small—so small that a very, very strong magnetic field is required to see any difference at all.
-
Nuclei that interact with magnetic fields are said to possess nuclear spin. The exact number of different energy levels a nucleus can adopt is determined by this nuclear spin, I, of the particular isotope. The nuclear spin I can have various values such as 0, 1/2, 1, 3/2 and the number of energy levels is given by 2I+ 1. Some examples are: 1H, I= 1/2; 2H (= D), I= 1; 11B, I= 5/2; 12C, I= 0.
-
The amount of energy needed to flip the nucleus can be provided by electromagnetic radiation of radiowave frequency. Radio waves flip the nucleus from the lower energy state to the higher state. The nucleus now wants to return to the lower energy state and, when it does so, the energy comes out again and this (a tiny pulse of radiofrequency electromagnetic radiation) is what is detected. The operation of a NMR machine is summarized below.
-
The sample of the unknown compound is dissolved in a suitable solvent and put in a very strong magnetic field. Any atomic nuclei with a nuclear spin now have different energy levels, the exact number of different energy levels depending on the value of the nuclear spin.
-
The sample is irradiated with a short pulse of radiofrequency energy. This disturbs the equilibrium balance between the two energy levels: some nuclei absorb the energy and are promoted to a higher energy level
-
The energy given out when the nuclei fall back down to the lower energy level is detected using a sophisticated radio receiver.
-
After lots of computational analysis involving complex mathematical operations, the results are displayed in the form of intensity (i.e. number of absorptions) against frequency. This is known as one dimensional NMR spectrum.
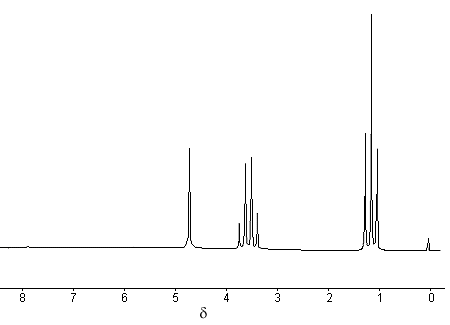
The proton NMR spectrum of ethanol is given below.