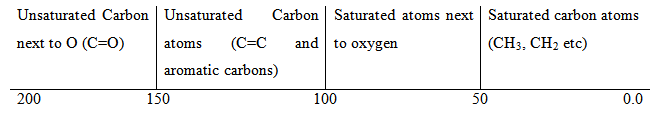- In the 1H NMR spectrum, it is also possible to obtain the number of protons that a particular signal corresponds to. Thus the signal at 0.98 corresponds to 3 protons for ethanol. This is because the area under each peak is directly proportional to the number of protons. This peak area is called integration value of the peak. Thus 1H NMR spectra can be quantitative in nature but the same cannot be said for 13C spectrum. For reasons beyond the scope of this text, they cannot be integrated to give the number of carbons.
- Similarly, the protons attached to particular type of carbon usually resonate in the same region. Thus, it is very useful to know the different regions on a 1H spectrum.
A similar region wise classification may be made for 13C NMR spectrum.
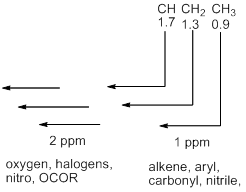
- If adding electronegative atoms are added to a carbon atom, electron density is progressively removed from it and the carbon nucleus and the hydrogen atoms attached to it are progressively deshielded. In general an oversimplified chart can be constructed regarding the values of chemical shifts in 1H NMR. However, it should be kept in mind that such a chart is purely a simplication of the situation and therefore exceptions are very common. These chemical shifts are additive. This means that if in a molecule there are two factors that should lead to an increase in the value of chemical shift of the proton concerned, then the additive effect due to these is to be considered.
• Methyl groups are to be started at 0.9, methylenes (CH2) at 1.3, and methines (CH) at 1.7 p.p.m. Any functional group is worth a one ppm downfield shift except oxygen and halogen for which the value goes two ppm downfield.