4.19.4.3 Artificial Base-Pairs Based on Hydrophobic Interactions- Replacement of Heterocyclic (Purine or Pyrimidine) Bases by Polycyclic Aromatic Hydrocarbons
Kool and colleagues have taken a different approach to develop novel fluorescent nucleotide base analogs for incorporation into nucleic acids. In their synthetic design, instead of modifying an existing heterocyclic nitrogenous-containing nucleoside base, they simply replace it with aromatic polycyclic hydrocarbon structures. Like the modified natural bases, these hydrocarbon aromatic structures can be incorporated with only small perturbation to the natural DNA structure and allow for close interactions including stacking within the DNA helix. In general, the hydrocarbon base moieties are designed to be non-polar, weakly hydrogen bonding and planar aromatic groups. Synthetic routes to 4-methyl-1H-indole, phenanthrene and pyrene fluorophores conjugated at the C(1) position of 2-deoxy-D-ribose (Figure 4.39) have been reported. In addition, the derivative with a pyrene moiety at C(1) in the α-D-form has been shown to be of use in DNA diagnostics strategies, where it efficiently forms excimers with neighboring pyrene labels. Pyrene derivatives in the β-D-form stabilize DNA helices markedly due to low polarity and have been incorporated into the DNA helix enzymatically by DNA polymerase. The synthesis of additional fluorescent aromatic hydrocarbon nucleoside analogs with novel fluorescence characteristics, like shifted ranges of excitation and emission wavelength maxima, has more recently been reported. These fluorophores included terphenyl, stilbene, terthio- phene, benzoterthiophene (Figure 4.39) and were conjugated to deoxyribose at the C(1) position in the α-D- form. The emission spectral maxima for the free nucleoside forms of these base analogs ranged from 345 nm to 536 nm, with fluorescence quantum yields that ranged from 0.025 to 0.67. Synthesis of the phosphoramidite derivative of each of these conjugated nucleoside base analogs was also reported, allowing straightforward incorporation using standard automated oligonucleotide synthesis. In addition to the conjugated fluorophore nucleosides, a C-nucleoside with cyclohexene at the 1- position was also synthesized as a non-fluorescent spacer. The rational for synthesizing this spacer was to attempt to disrupt ideal stacking conditions that results in a high degree of quenching of the aromatic hydrocarbon base analogs conjugated at C(1) when incorporated into oligonucleotides.
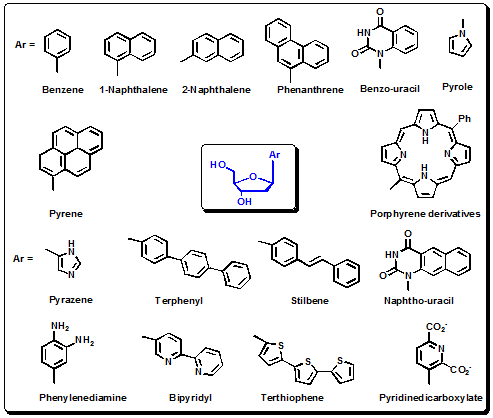
Figure 4.39: Structures of a series of polycyclic aromatic planar hydrocarbon and oligothiophene base analogs that display a range of fluorescent properties: