Pteridineadenine analogs:Pteridines are a class of bicyclic planar compounds that are structurally similar to purines. Two pteridine adenine analogs, 4-amino-6-methyl-8-(2-deoxy-β-D-ribofuranosyl)-7(8H)-pteridone (6-MAP) and 4-amino-2,6-dimethyl-8-(2’-deoxy-β-D-ribofuranosyl)-7(8H)-pteridone (DMAP ) (Figure 4.31) have also been synthesized and phosphoramidite also incorporated into DNA oligonucleotides for fluorescence measurements. Like the pteridine guanine analogs, these analogs display significant quenching of fluorescence intensity, increased complexity of decay curves and decreased mean fluorescence lifetimes when incorporated into oligonucleotides. The pteridine analogs have been shown to be minimally disruptive of DNA structure as evidenced by the similarity of melting point temperatures of pteridine-containing oligonucleotides with unmodified control oligonucleotides.
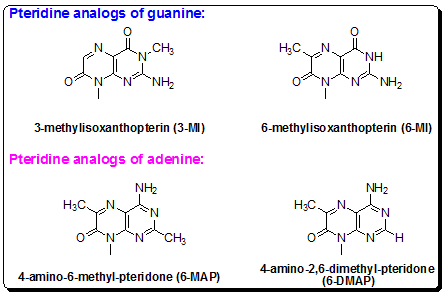
Figure 4.31: (5). Two pteridine analogs of guanine, (A) 3-MI and (B) 6-MI and two adenine analogs synthesized from the pteridine class of compounds, (C) DMAP and (D) 6-MAP, which have been incorporated into short DNA oligonucleotides for structural and enzymatic studies.
4.19.4.1.2.2. Guanine Base Analogs
Pteridine Guanine Analogue: Hawkins and colleagues have described the synthesis of several pteridine base analogs, some of which are highly fluorescent with ΦF's ranging from 0.03 to 0.88. Among the available pteridines, two guanine analogs, 3-methylisoxanthopterin (3-MI) and 6- methylisoxanthopterin (6-MI) have been synthesized as phosphoramidites and incorporated into DNA oligonucleotides. Like 2-AP, incorporation of these probes into oligonucleotides significantly quenched their fluorescence signal and the degree of quenching correlated with the number and proximity of purines in the oligonucleotide. Incorporation also resulted in a shift in absorbance, emission and decay-associated spectra for 6-MI. An increase in the complexity of the fluorescence decay curve and a decrease in the mean lifetime were also observed for both the 3-MI and 6-MI probes. Formation of double stranded oligonucleotides did not substantially increase the degree of quenching but generally increased the complexity of decay curves and decreased mean fluorescence lifetimes.
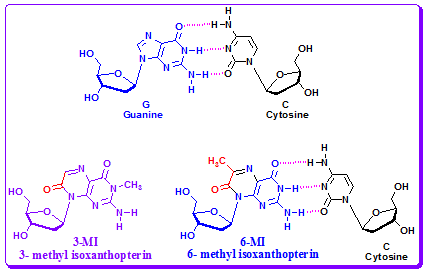
Figure 4.32: 3-MI and 6-MI as pteridine guanine analogues
4.19.4.1.3. Pyrimidine base Analogues
4.19.4.1.3.1. Thymine Analogue
Benzo[g]quinazoline based thymine base analogs: Godde and colleagues have synthesized bases with extended aromatic domains that increase third strand binding through stacking interactions. One of the polycyclic aromatic base analogs of thymine, benzo[g]quinazoline-2,4-(1H,3H)-dione (Figure 4.33), was found to display strong fluorescence emission centered at 434 nm (ΦF ~ 0.82) and two major excitation maxima (260 and 360 nm). Formation of the triple helical structure using a third oligopyrimidine Hoogsteen strand that contained this fluorescent thymine analog, resulted in a shift of the fluorescence emission maximum to shorter wavelengths and a decrease in fluorescence intensity. In a duplex, it did not produce any significant changes in fluorescence properties. Thus, the sensitivity of this base analog to the helical conformation allowed selective detection of triplex over duplex formation.
Thymine analogue: Another thymine analog 5-methyl-2-pyrimidinone (Figure 4.33) has been synthesized and used in early studies of DNA duplexes. This base analog does not pair well with adenine, however, using time-resolved fluorescence decay measurements it was shown that the predominant state of the base in the context of a DNA oligonucleotide is stacked so that its fluorescence is efficiently quenched.
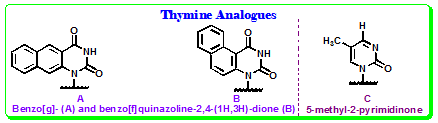
Figure 4.33: benzo[g]/[f]quinazoline-2,4-(1H,3H)-dione and 5-methyl-2-pyrimidinone as Thymine analogue