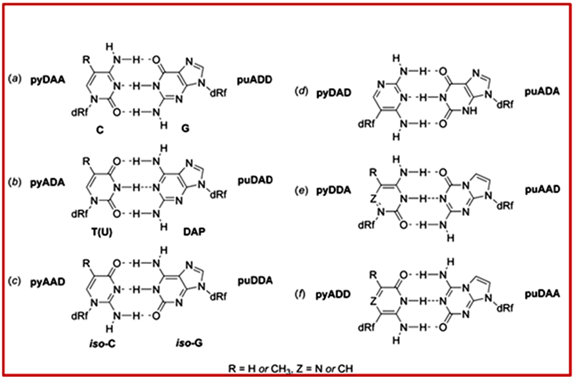
Figure 4.28: Several base pairs with H-bond
4.19.4.1.2. Purine base Analogues
4.19.4.1.2.1. Adenine Base Analogs
2-aminopurine (2-AP) as Adenine Base Analogue:More than 30 years ago, Stryer and colleagues first demonstrated that 2-AP (Figure 4.29), a structural isomer of adenine, was strongly fluorescent. Being structurally similar to adenine (6-aminopurine) 2-AP is a non-perturbing substitution and form thermodynamically equivalent base pairs with thymine in DNA helices and uracil in RNA helices. In contrast to adenine, 2-AP is also capable of forming a base pair with cytosine at a substantially higher frequency, which is the basis for 2-AP’s mutagenicity. The introduction of 2-AP into both DNA and RNA oligonucleotide sequences in a site-specific manner is straightforward and the quantum yield of 2-AP is highly sensitive to its microenvironment and insensitive to base pairing and other H-bonding interactions, thus, allows to be used to detect subtle conformational changes in nucleic acids.
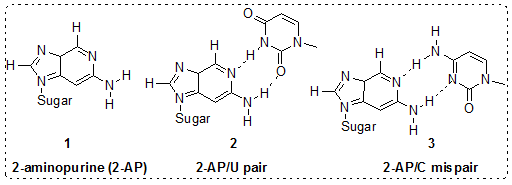
Figure 4.29: (a) Structure of the highly fluorescent nucleotide base analog 2-aminopurine (2-AP), 1; 2-AP-uracil pair, 2 and 2-AP-cytosine mispair, 3.
In 1988, Andre Chollet et al., have introduced the base analogue 2-aminoadenine (2,6-diaminopurine, D) at selected positions into synthetic oligodeoxyribonucleotides and DNA by the combined use of chemical and enzymatic methods. 2-aminoadenine substitution for adenine introduces changes in the minor groove of DNA and creates an additional hydrogen bond in the Watson-Crick base pair with thymine. Oligonucleotide hybridization probes containing 2-aminoadenine showed increased selectivity and hybridization strength during DNA- DNA hybridization to phage or genomic target DNA. Properties of the base analogue with respect to DNA modifying enzymes were examined. 2-aminoadenine was used to probe minor groove determinants during the treatment of DNA by 12 restriction endonucleases. Inhibition of cleavage was found for several restriction enzymes.
Recently, Tom Brown, has synthesized the heterocyclic base 7-aminopropargyl-7-deaza-2,6-diaminopurine (D) and incorporated into oligodeoxynucleotides. They have shown that D:T has similar thermodynamic stability to G:C and is a stable analogue of A:T.
ATP/AMP Analogs: Other fluorescent analogs of adenine have been developed and used primarily as probes of AMP/ATP binding by enzymes (Figure 4.30). These other analogs include etheno-ATP and lin-benzo-AMP, which are analogs of ATP and AMP, respectively. Another ATP analog, formycin 5'-triphosphate, has been used as a substrate analog for adenylate cyclase. These analogs have been used primarily in studies of nucleotide cofactor binding to enzymes.
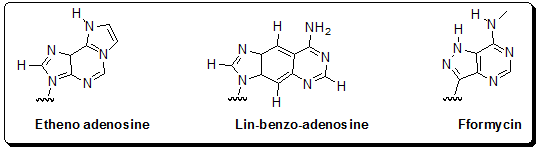
Figure 4.30: Structures of adenine base analogs used to construct fluorescent nucleoside and nucleotide analogs- Etheno-adenosine, lin-benzo-adenosine, and formycin.