Table 1: Bond Lengths for 1-Alkyl-3-methylimidazolium Cations from QM Calculations and X-ray Diffraction Data [a: Holbrey et al., 2003a ; b: Holbrey et al., 2003b]
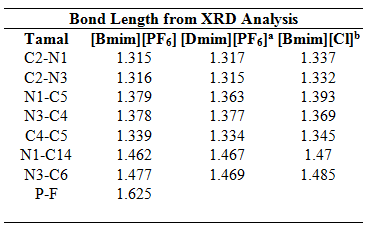
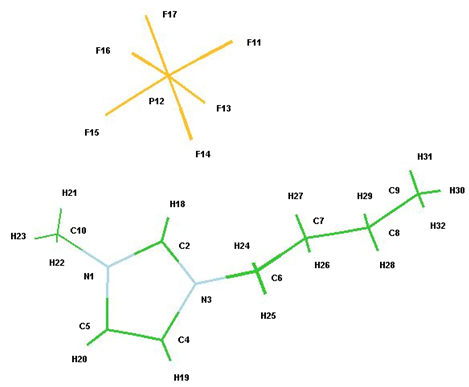
Figure 6.: Optimized Structure for [Bmim][PF6] using HF/6-31G*
For calculating the partial charges the following command needs to be called in GAUSSIAN03 package:

The pop(npa,chelpg) creates the population containing the list of partial charges of all atoms.
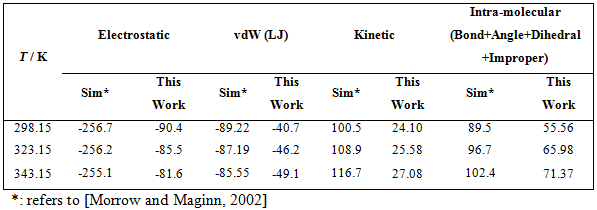
Benchmarked properties include the molar volume, volume expansivity and isothermal compressibility. Simulations are carried out at T = 298.15, 323.15 and 343.15 K and 1 atm pressure. A break down of the various forms of potential energies is given in Table 2.This includes the coulombic, van der Waals (vdW), intramolecular and the kinetic energies. It can be seen that the electrostatic energies are approaching twice as compared to the intramolecular energy; this shows that the ionic liquids are more sensitive to the electrostatic portion of the force field, thereby rightly being called as “ionic liquids”.
Table 2. Validations of Energies of various forms (all units in kJ/mol)