STEP II: Minimization of Initial Structure
The first step in preparing the system for production of MD simulations involves energy minimization using conjugate gradient method. The purpose of this stage is to adjust the geometry to the force field, distribute the molecules .Further this step also helps in avoiding the steric clashes created by guessing coordinates of atoms during generation of psf file.
STEP III: Heating of the System
During this stage the temperature of the system is linearly increased from 0 K to the required temperature within a specified time. At each integration step velocities are drawn from a Maxwell distribution and the temperature is incremented by 0.01 K. (Fig 3).
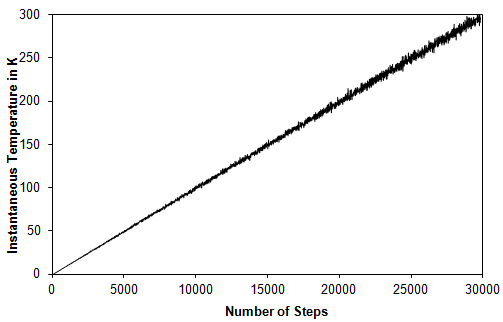
Figure 3: Increase in the instantaneous temperature with number of steps
STEP IV: Equilibration of the System
The purpose of equilibrium stage is to enable the system to evolve from the starting configuration to the equilibrium state. In this stage certain thermodynamic properties are monitored such as energy, temperature and pressure.So equilibriation stage is done in an NVT ensemble and is thus used to equilibriate kinetic and potential energies, i.e., to distribute the kinetic energy “inserted” into the system during heating among all degrees of freedom. This means that the potential energy “falls behind” and must be equilibrated with the kinetic energy. As soon as potential energy levels off the equilibration stage is completed. Fig. 4 shows the temperature and the potential energyas a function of time (Fig 5). After initial rapid decrease from about -8000 kcal/mol the potential energyfluctuates near a constant level. This behavior suggests that the system is equilibriated on a timescale much shorter than 30 ps.
The potential energy in Fig. 7 levels off at about -9000 kcal/mol, whereas the final energy during heating stage is somewhat less (-9600 kcal/mol). It should be noted that the velocities are rescaled after each integration step to the desired temperature. Total number of steps taken is 32300.Each time step here is of 1 femtosecond.
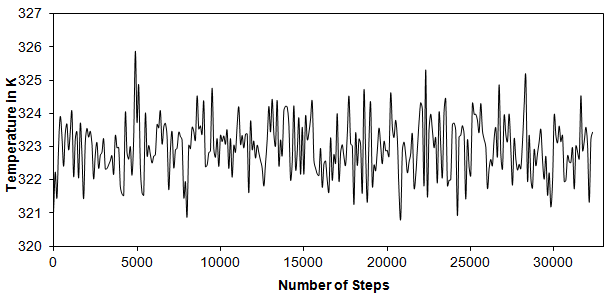
Figure 4: Time Dependence of instantaneous temperature T = 323.15 K
(Root Mean Square Error: = 0.803 K)