A typical process flow diagram for isomerization of C5 and C6 alkanes to branched products is shown in Fig 3. The process conditions of C5 /C6 isomerization depend on the activity of the catalyst as well as on the composition of the feed. For example, feed rich in hexane requires high hydrogen pressure (~35-100 atm) to maintain low coke formation where as pentane rich feed can be processed at lower pressure (less than 27 atm) at temperatures between 120- 175°C. The feed is mixed with hydrogen to minimize the coke formation and passes through the reactor followed by separator. The hydrogen from separator is recycled. The product is further stabilized by removing lighter gases. The products are separated from the reactants to maintain a favorable equilibrium and the unreacted normal hydrocarbons are recycled. Any isopentane retained in feed or recycle is immediately separated at the front end of the process as shown in the figure.
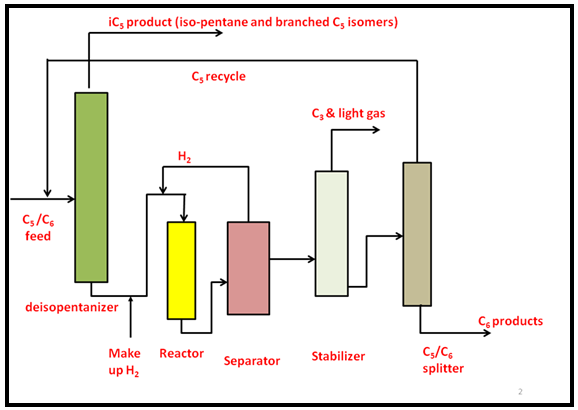
Fig. 3. Process flow diagram for isomerization of C5 and C6 alkanes to branched products
Catalysts
Initially Pt supported on Al2O3– SiO2 were extensively used as isomerization catalysts. More recently, Pt- ZSM5 zeolites have proved to be better catalyst. Pt centers dehydrogenate the alkanes while acid sites isomerize the resultant alkenes which are then rehydrogenated by Pt to isomerized alkanes. Pt also serves to moderate coke formation by hydrogenation of coke precursors.
Mechanism and kinetics
For isomerization of C4- C8 hydrocarbons occurs through carbenium ion intermediate. Both, empirical power rate law and Langmuir–Hinshelwood type of kinetic models have been used to model the kinetics.
Catalysts deactivation
Catalysts are mainly deactivated by coke deposition. Coke deposition can be minimized by increasing H2 pressure. The catalysts can be regenerated by controlled burning of coke. Other poisons such as sulfur compounds, water and traces of aromatics can also be removed during coke burn off. The adsorbed sulfur and water on the catalyst can also be removed by decreasing their partial pressures in the stream, when they are reversibly adsorbed.