Mechanism and kinetics
Reforming involves simultaneous occurrence of several reactions thereby making mechanism of reforming very complex. Lumped kinetic models are used in which naphtha components are lumped into functional groups i.e. alkanes, naphthenes and aromatics. Some of the reaction scheme used to develop catalytic reforming kinetic model is shown in Fig.2
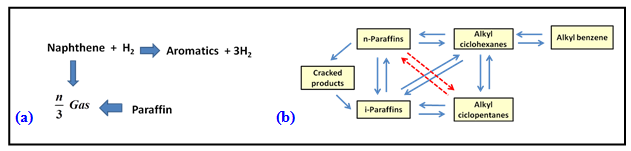
Fig.2. Reaction scheme for catalytic reforming [1]
Deactivations
Major sources of deactivation are:
– Coke formations
– Sintering
– Poisoning by S,N compounds
– Poisoning by Arsenic, Lead, other metals (Cd, Cr, Co, Fe etc.); As< 1ppb; Pb< 20 ppb; other metals < 600 ppm
Coke formation is highest in last reactor. The feed is hydrotreated to remove sulfur and nitrogen compounds.
Isomerization
Isomerization involves conversion of less useful hydrocarbons to their corresponding more desirable isomers. For example, normal butane, pentane or hexane can be isomerizd to their branched isomers having higher octane number. Similarly, m-xylene can be isomerized to para-xylene (p-xylene) which is used as feed for production of pure terephthalic acid. Consequently, out of all the xylenes, p-xylene has the largest commercial market.
Reactions & Process conditions
The isomerization reactions of butane, pentane and hexane are as follows.
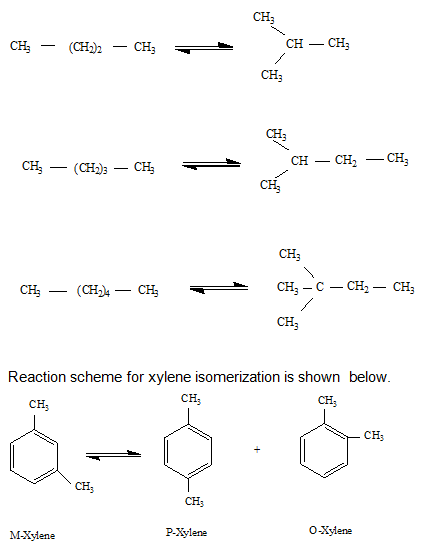
Isomerization of xylene is carried out at 400-4500C and 10-20 H2 atm over platinum based catalysts and equilibrium distribution of different xylene isomers is obtained. The branched products of the isomerization reactions are thermodynamically more favorable at lower temperature. The most active catalysts are strong acids such as anhydrous AlCl3 which are also corrosive. Solid catalysts such as Pt/Cl− /Al2O3 are also used although have lower activity at lower temperature.