Process
Naphtha reforming involves different types of reactions which are thermodynamically and kinetically favored at different reaction conditions. For example, dehydrogenations are favored thermodynamically and kinetically at high temperature and low pressure while isomerization is kinetically favored at high temperature and pressure. The high temperature can cause excessive dehydrogenation especially of aromatics in the feed leading to high rates of formation of coke. To minimize the coke formation, the hydrogen is added in large excess to feed. The naphtha feed is initially hydrotreated to remove sulfur and nitrogen compounds as the reforming catalyst are sensitive to these compounds. The naphtha reforming process is therefore carried out in series of different reactors to optimize various reactions. Typically the process involves three reactor setups. The 1st reactor is designed for dehydrogenation of naphthenes to aromatic. The 2nd reactor is designed for additional dehydrogenation with isomerization and the 3rd reactor is for dehydrocyclization reactions which are kinetically least favorable and therefore require large bed volume. The reactors are loaded with increasing amount of catalyst and the last reactor contains the largest amount of catalyst. Depending on feed the commercial reformers are operated at 5-35 atm and 450-500°C Due to endothermic reactions, the exit stream from each adiabatic reactor needs to be reheated to 5000C before entering the next reactor.
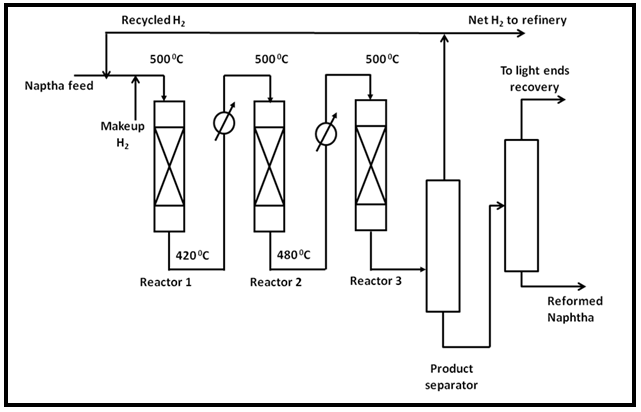
Fig. 1. Schematic diagram for naphtha reforming process
Catalysts
Reforming catalysts involve dual functions; acidity and dehydrogenation function. The acidic center of the catalyst promotes the structural changes involved in isomerization reactions. The dehydrogenation reactions are catalyzed by metal site of the catalyst. Reforming catalyst usually contains Pt as the metal component and modified γ-Al2O3 as the source of acidity. The acidity of the Al2O3 is further enhanced by incorporation of chloride ions into its structure. Addition of small amount of Re along with pretreatment with H2S improves the life of the catalyst. Resulfide breaks up the Pt metal surface into smaller entities and decreases the probability of coke formation. Other promoter such as Ge, Tr and Sn have also been used in place of Re depending upon feed or process condition.