Case II: Consider a reaction in a mixed reactor under changing flow condition so that concentration is constant. Let parallel deactivation occurs. Assume the main reaction to be nth order and the deactivation reaction to be of first order in activity ‘a'.
Parallel deactivation 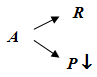
Rate equation Deactivation rate |
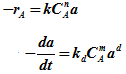 |
........................... ......................... |
Since concentration is constant,
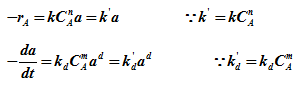
For mixed reactor,
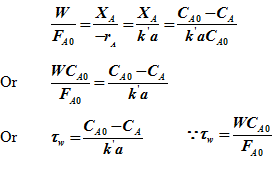 |
(9) |
Now , ![]()
For first order activity, d =1
Again, ![]()
Then substituting ‘a' in equation (9)
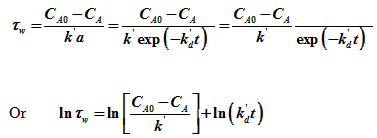
Plotting ![]() versus ‘t' gives a straight line of slope of kd and intercept
versus ‘t' gives a straight line of slope of kd and intercept 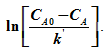
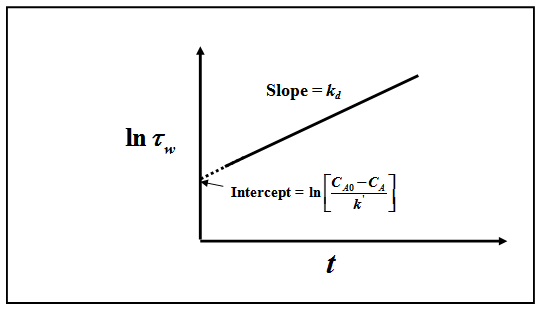
Fig. 2 . Plot to determine rate constant and deactivation rate constant for a reaction over deactivating catalysts for mixed reactor, changing flow condition and parallel deactivation
Book Reference :
• O. Levenspiel , Chemical reaction engineering, John Wiley & sons. 1995
• R. J. Farrauto & C. H. Bartholomew, Fundamentals of Industrial catalytic Processes, Blackie Academic & Professional, 1997
• H. S. Fogler, Elements of Chemical reaction engineering , Prentice Hall of India, 1999
• J. M. Smith, Chemical Engineering Kinetics , McGrawHill Book Company, 1981
• J.J. Carberry , Chemical and catalytic reaction Engineering, Dover Publications, 2001