Deactivation kinetics
Activity of catalysts pellet at any time is defined as
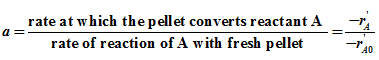
Reaction rate can be considered as function of temperature, reactant concentration and present activity of the catalysts pellet. Therefore for n th order reaction, rate can be written as
![]()
Where a is the present activity of the catalysts.
Similarly deactivation rate can be considered as function of temperature, concentration of substance that deactivates the catalysts (which can be reactant, product or impurities) and present activity of the catalysts pellets. Hence deactivation rate can be given as,
![]()
i = A (reactant),R (product) ,P(3rd substance)
Here d is called order of deactivation, kd is deactivation rate constant and n΄ is the concentration dependency.
Deactivation reaction rate for different types.
1. Parallel , ![]()
Reaction rate is ![]()
| Deactivation rate is |
d = order of deactivations |
2. Series ![]()
Reaction rate is ![]()
Deactivation rate is ![]()
3. Side-by-side deactivations , ![]()
Reaction rate is ![]()
Deactivation rate is ![]()
4. Independent deactivations
Reaction rate is ![]()
Deactivation rate is ![]()