Mechanism of catalysts deactivation
The deactivation of catalysts can occur by different mechanism as explained below:
1. Parallel deactivation
Reactant (A) produces a side product (P) which can deposit on the surface thereby deactivating it. Deposition depends on reactant concentration.
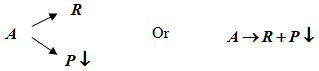
2. Series deactivations
Reaction product (R) can decompose or reacts further to produce material (P) that deposit and deactivate the surface. In this case deposition depends on product concentration.
3. Side-by-side deactivations
In this mechanism, impurity in feed (P) deposits on the surface deactivating it. Deposition depends on concentration of impurities in the feed.
4. Independent deactivations
This catalyst decay involves structural modifications or sintering of catalysts surface caused by exposure of catalysts to extreme conditions such as high temperature. This decay is dependent on time that the catalyst spends in high temperature conditions and is independent of the materials in the fluid.