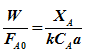Deactivation kinetics for poisoning
or petroleum feed containing trace impurities such as sulfur, the poisoning reaction can be written as ![]()
The rate of deactivation is given as
![]()
The C p is the concentration of poison in feed. The ‘m' and ‘q' are respective orders.
The rate of removal of poison from the reactant by catalyst sites is proportional to the number of sites that are not poisoned (Cto - Cps) and concentration of poisons in the gas phase, Cp. The Cps is the concentration of poisoned sites and Cto is the total number of fresh sites initially available. Then rate of removal of poison is equal to rate of formation of poisoned site and is given as
| (4) |
Dividing throughout by Cto and if ‘f' is the fraction of the total number of the sites that have been poisoned then equation (4) can be written as
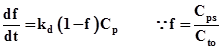 |
(5) |
The fraction of sites available for adsorption (1-f) is essentially the activity a (t). Then equation (5) becomes
![]()
Determination of deactivation kinetic parameters
Case 1: Consider a reaction in a mixed reactor under constant flow condition. Let independent deactivation occurs. Assume the main reaction to be 1st order and the deactivation reaction to be also in first order in activity ‘a'
Then
| Reaction rate |
(6) |
| Deactivation rate |
(7) |
Integrating equation (7)
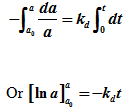 |
|
. a0 is initial activity |
......................
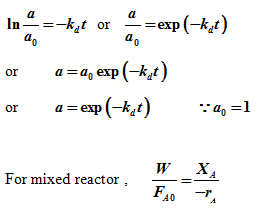
Substituting expression for rate from equation (6)