Deactivation kinetics for sintering
Most commonly used decay law for sintering is second order with respect to present activity.
![]()
The kd is the sintering decay constant. Integration with a=1 at time t=0 gives
| (1) |
Activity can be measured in terms of amount of sintering. If the total active surface area of the catalyst is Sao at t = 0 and Sa at any time t, then activity can be written as
| (2) |
From equation (1) and (2)
![]()
The sintering decay constant kd is given as
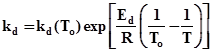
Ed is the decay activation energy. For reforming of heptanes on Pt /Al2O3 value of Ed is in the order of 70 kcal/mol.
Deactivation kinetics for coking or fouling
Various empirical relations are available for calculation of amount of coke deposited on catalyst surface after a time t.
| (3) |
Cc is the concentration of carbon on the surface in g/m2. The ‘n' and ‘A' are fouling parameter functions of feed rate. Representative value of A and n for cracking of light gas oil is 0.47 and 0.5 respectively when ‘t' is in minute.
The activity can be related to amount of coke deposited on the surface by following relation:
![]()
Substituting value of Cc from relation (3), in term of process time, activity can be expressed as shown below.
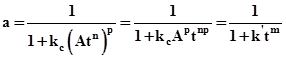
or
![]()
Other dimensionless fouling correlations are also developed by several groups.