Where for spherical pellets of radius rs , ![]() for a first-order reaction is given as
for a first-order reaction is given as
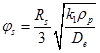 |
(13) |
k1 is rate constant, ρp is pellet density; De is effective diffusivity.
Hence, to calculate Thiele modulus, both effective diffusivity De and rate constant k1 need to be known. Fig 3 shows the typical plot of η vs ![]() .
.
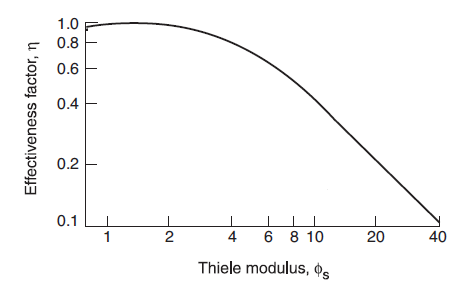
Fig. 3. Typical plot effectiveness factor η as function of Thiele modulus φs
As the figure shows, for small values of ![]() . Equation (13) shows that small values of
. Equation (13) shows that small values of ![]() are obtained when the:
are obtained when the:
- 1. pellets are small
2. diffusivity is large
3. reaction is intrinsically slow ( catalysts of low activity).
For ![]() ,
, ![]() can be approximated as
can be approximated as ![]() . For such large
. For such large ![]() , intra-particle diffusion has a large effect on the rate. At this condition, diffusion into the pellet is relatively slow and reaction occurs before the reactant has diffused far into the pellet. When
, intra-particle diffusion has a large effect on the rate. At this condition, diffusion into the pellet is relatively slow and reaction occurs before the reactant has diffused far into the pellet. When ![]() , rate for the whole pellet is the same as the rate at surface concentration Cs and the entire catalyst surface is fully active.
, rate for the whole pellet is the same as the rate at surface concentration Cs and the entire catalyst surface is fully active.