Effectiveness factor at isothermal conditions
For an irreversible first order reaction A → B
, at isothermal condition the rate at surface condition can be written as ![]() . Then the actual global rate at outer surface condition,
. Then the actual global rate at outer surface condition, ![]() .
.
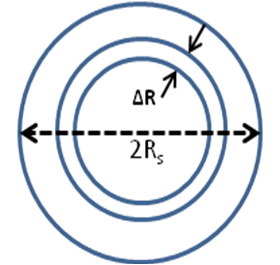
For a spherical catalyst pellet of radius ‘Rs', mass balance over a spherical shell volume of thickness ![]() at steady state will be:
at steady state will be:
(Rate of diffusion of reactant into element) – (Rate of diffusion of reactant out of element) = Rate of disappearance of reactant within element due to reaction
Rate of reaction per unit mass of catalyst = ![]()
Rate of reaction per unit volume of catalyst = ![]() .
.
Where ![]() is the density of pellet.
is the density of pellet.
Then the mass balance equation over the element of thickness ![]() can be written as
can be written as
![]()
If limit is taken as ![]() and assuming that effective diffusivity is independent of concentration of reactant the above equation can be written as
and assuming that effective diffusivity is independent of concentration of reactant the above equation can be written as
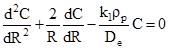 |
(7) |
Boundary conditions:
| At the center of the pellet, | (8) |
| At outer surface | (9) |
Solving the differential equation (7) with boundary conditions (8) and (9) following relation is obtained
| (10) |
Where ![]() is a dimensionless group defined as
is a dimensionless group defined as
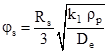 |
|