Assume that for a gaseous irreversible reaction A → B, the reaction and diffusion in a single cylindrical pore results in equimolal counter diffusion, or NB = - NA. Then for reactant A, the combined diffusivity in a cylindrical pore is given as
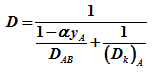 |
(1) |
where ![]() is the bulk diffusion contribution and
is the bulk diffusion contribution and ![]() is the Knudsen diffusion contribution.
is the Knudsen diffusion contribution.
Here 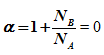 = 0 as NB = - NA
= 0 as NB = - NA
Or 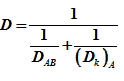 |
(2) |
When pore radius is large ![]() , = bulk diffusion. If pore radius is very small, Knudsen diffusivity becomes is controlling and
, = bulk diffusion. If pore radius is very small, Knudsen diffusivity becomes is controlling and ![]()
Bulk diffusivity at moderate temperature and pressure can be evaluated using Chapman –Enskog equation. For binary gas mixture of A and B
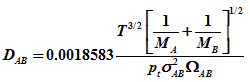 |
(3) |
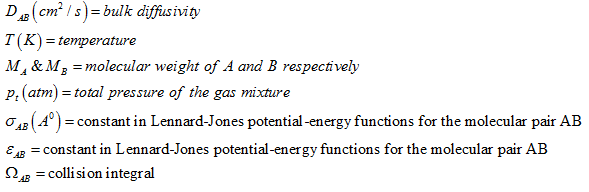
Knudsen diffusivity can be evaluated by the following equation:
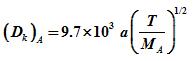 |
(4) |

The diffusivity of A in multicomponent system containing n components is given as
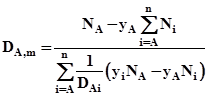
Here Ni is taken as positive if diffusion is in same direction as that of A and negative if in opposite direction. The DAi Ai are the binary diffusivities of component A with respect to component ‘i'. The yA is molfraction of A and yi molfraction of ‘i'