Random pore model
This model considers that pellets consist of both macro and micro pores. Pellets consist of assembly of small particles containing micropores and the void space between the particles constitutes the macropores. Transport in the pellet is assumed to occur by combination of diffusion through the macro-regions having void fraction εM and the micro regions having void fraction εμ. The magnitude of individual contribution is dependent on their effective cross-sectional areas perpendicular to the direction of diffusion.
The resultant expression for effective diffusivity De is given as
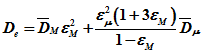 |
(6) |
![]() and
and ![]() are combined diffusivity in macropores and micropores respectively and obtained by applying equation for combined diffusivity D to macro and micro regions as follows.
are combined diffusivity in macropores and micropores respectively and obtained by applying equation for combined diffusivity D to macro and micro regions as follows.
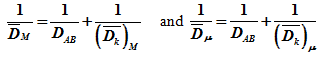
No tortuosity factor is involved in this model. The actual path length is equal to the distance co-ordinate in the direction of diffusion.
For a pellet containing only macropores, substituting ![]() in equation 18 gives
in equation 18 gives ![]() .
.
Similarly for materials with only micropores, putting ![]() in equation 18 gives
in equation 18 gives ![]()
Effectiveness factor η
In a porous catalyst, the internal surface area within the pores constitutes the effective surface on which the active metals are deposited. Hence, for reaction to occur, the reactants need to travel through the pores to reach the active sites. If there is any resistance in diffusion of the reactants through the pores, then both the activity and selectivity of the process is affected. In the absence of any internal mass transfer resistance within the pores the concentration of reactant s can be assumed to uniform and equal to the surface concentration. Hence, the rate of reaction will also be uniform across the catalyst pellet and equal to the rate evaluated at surface conditions. However, in the presence of diffusional resistance, concentration of reactants gradually decreases from the surface towards the center of the catalyst pellet, resulting in a concentration gradient. When this concentration gradient is large enough, significant variation in reaction rate occurs within the pellet. The average rate within pellet will be less than the rate corresponding to the surface concentration. As pellet size decreases, the effect of intrapellet transport becomes less significant.
Rate of reaction for the whole catalyst pellets at any time is defined with respect to outer surface conditions in term of effectiveness factor η as:

Actual rate of whole pellet rp takes intrapellet mass transfer effects if any into account.