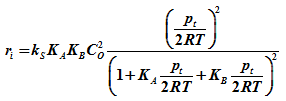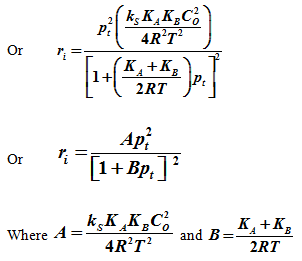Initial rate analysis
Initial rate analysis for a reaction may be done to understand the mechanism of a reaction. Initial rate is defined as the rate at zero conversion. It is obtained by extrapolation of the rate vs conversion data to zero conversion. For higher accuracy, the conversion data are collected at very low conversion region. In this method, the dependence of initial rate data on partial pressure or total pressure of the reactant is studied.
Consider the reaction ![]()
The rate expression derived from Langmuir Hinshelwood model when surface reaction is controlling is
 ........................(2)
........................(2)
Now at zero conversion all the product concentration will tend to zero, hence putting
CC =0 & CD =0, the rate expression [2] simplifies to
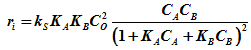 .....................................................(3)
.....................................................(3)
Where ri is the initial rate. Now if a equimolal initial concentration of CA and CB is used then CA= CB. Then assuming ideal gas mixture, ![]() , where pA is the partial pressure of A.
, where pA is the partial pressure of A.
Similarly ![]() where pB is the partial pressure of B .
where pB is the partial pressure of B .
Now since it is an equimolal mixture, then at initial condition. ![]() where pt is the total pressure.
where pt is the total pressure.
Or ![]()
Then substituting CA and CB in equation [3] becomes