Analyzing data from laboratory reactors
The objectives of analyzing data include
- • Determining catalyst activity, selectivity and stability
• Determining the effect of important process variables such as temperature, pressure, reactant concentrations
• Finding a rate equation
Elimination of mass transfer and pore diffusion limitations
In a solid catalyzed reaction, the presence of mass transfer and pore diffusional resistances can significantly affect the rate of the reaction. The details will be discussed in lecture no. 22 to 24. To ensure that no internal or external mass transfer resistances are present under the operating conditions, preliminary experiments are carried out.
Elimination of mass transfer limitations
For checking if external mass transfer is affecting the rate of reaction, conversion data is obtained by increasing the flow rate of the feed but keeping the W/FAO constant. Fig. 5 shows the plot of conversion as a function of linear velocity of fluid at constant W/FAO at a given temperature, usually the highest temperature is used during reaction.
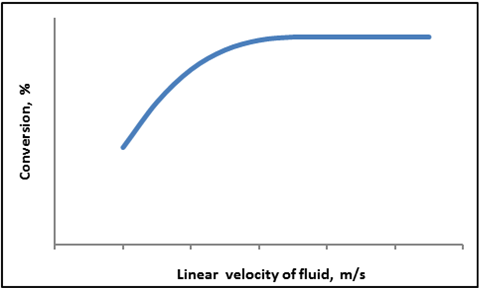
Fig. 5. Typical plot of conversion as a function of linear velocity of fluid at constant W/FAO and temperature
It can be observed that at low fluid velocity, conversion increases with increasing fluid velocity and subsequently becomes constant at higher flow rates, implying that external mass transfer does not affect the rate of reaction at this fluid velocity. Due to external mass transfer limitation the concentration of reactant near catalyst surface is lower than the bulk phase concentration. The effect is maximum at lower fluid velocity when the resistance to transport is considerable. With increase in fluid velocity, the turbulence effectively enhances the mixing in fluid phase and the concentration of reactant near the catalyst surface approaches the bulk phase concentration. Consequently reaction rate increases increasing the conversion. At high velocity due to high turbulence the mass transfer limitation is totally removed and the reactant concentration near the catalyst surface becomes equal to that of bulk phase concentration and the conversion reaches the maximum limiting values. Thereafter any change in fluid velocity does not affect the conversion as shown in the figure. Kinetic study should always be carried out at conditions where the effect of external mass transfer on conversion is negligible.
Elimination of pore diffusion limitations
Weisz criterion 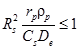 can be used to determine the significance of intraparticle diffusion. Here Rs = pellet radius; rp = observed rate; ρp = pellet density, Cs = surface concentration; De effective diffusivity. Reactions should be carried out at the conditions when Weisz criterion is satisfied so that the intraparticle diffusion could be neglected for these operating conditions. This is discussed in details in lecture no. 24. The effectiveness of porous catalyst can also be determined experimentally by measuring conversion with successively smaller catalyst pellets until no further change in conversion occurs. The pore diffusion resistance decreases the concentration within the pellets thereby decreasing the conversion. The pore diffusion resistance is considered to be negligible for the catalyst size range for which the conversion does not change with size. For study of intrinsic reaction kinetics, catalyst pellet size is chosen where the intraparticle diffusion could be neglected.
can be used to determine the significance of intraparticle diffusion. Here Rs = pellet radius; rp = observed rate; ρp = pellet density, Cs = surface concentration; De effective diffusivity. Reactions should be carried out at the conditions when Weisz criterion is satisfied so that the intraparticle diffusion could be neglected for these operating conditions. This is discussed in details in lecture no. 24. The effectiveness of porous catalyst can also be determined experimentally by measuring conversion with successively smaller catalyst pellets until no further change in conversion occurs. The pore diffusion resistance decreases the concentration within the pellets thereby decreasing the conversion. The pore diffusion resistance is considered to be negligible for the catalyst size range for which the conversion does not change with size. For study of intrinsic reaction kinetics, catalyst pellet size is chosen where the intraparticle diffusion could be neglected.