Method 2 : Preparing buffer with NaH2PO4 and Na2HPO4
NaH2PO4 and Na2HPO4-7H2O completely dissociate in water to provide conjugate acid ![]() and conjugate base
and conjugate base ![]() . As is evident from the Henderson-Hasselbalch equation (equation 3.8), the pH of the solution is determined by the ratio of conjugate base to the conjugate acid. Let us calculate this ratio:
. As is evident from the Henderson-Hasselbalch equation (equation 3.8), the pH of the solution is determined by the ratio of conjugate base to the conjugate acid. Let us calculate this ratio:
(3.8) |
For the given pair of conjugate acid and base, pKa2 of phosphoric acid is to be used:
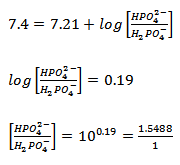 |
(3.20) |
Therefore, Na2HPO4-7H2O: NaH2PO4 ratio for preparing the required buffer is 1.5488:1. For preparing one liter of buffer, the required number of moles of phosphate species = 0.2 i.e. 200 mmoles .

Let us now see how to prepare the buffer:
- Weigh accurately 4.68 g of NaH2PO4 and 16.35 g of Na2HPO4-7H2O.
- Transfer the salts into a 1 liter conical flask and add 400 ml distilled water.
- Shake the flask well to achieve complete dissolution of the salts
- Wash the electrode of the pH meter thoroughly with distilled water.
- Place the pH electrode into the phosphate solution and measure its pH.
- Although the amounts of the conjugate acid and conjugate base are weighed so as to achieve a pH of 7.4, the pH of the solution is seldom 7.4.
- Although the amounts of the conjugate acid and conjugate base are weighed so as to achieve a pH of 7.4, the pH of the solution is seldom 7.4.
- If the pH of the solution is <7.4, add 1 N NaOH dropwise, shake the flask well and measure the pH. Keep doing this until a pH of 7.4 is achieved.
- If the pH of the solution is >7.4, add 1 N H3PO4 dropwise, shake the flask well and measure the pH. Keep doing this until a pH of 7.4 is achieved.
- Addition of H3PO4 increases the concentration of a phosphate species in the solution; you will therefore end up with a buffer that will have a pH of 7.4 but the concentration of the buffer will be slightly more than 200 mM .