Again, 0.1 – x can be approximated to 0.1 and 0.05 + x can be approximated to 0.05 . Equation 3.12, therefore, reduces to:
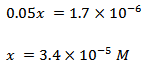 |
(3.15) |
Therefore, the equilibrium concentrations of CH3COOH, CH3COO−, and H3O+ in the presence of 0.5 M sodium acetate are ~99.966 mM , 0.084 mM , and 0.034 mM , respectively.
In case, x is not small compared to the concentration of the acid and the common ion provided by the solute, the quadratic equation needs to be solved. Let us take equation 3.14:
(3.17) |
Here we see that ![]() equation 3.17 therefore reduces to:
equation 3.17 therefore reduces to:
(3.18) |
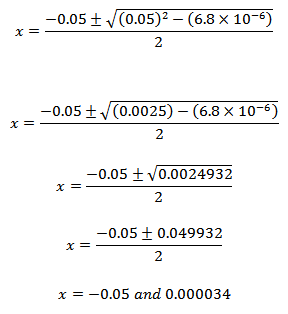 |
|
As concentration cannot be negative, x = 3.4×10-5. The result is same as obtained in equation 3.16. This shows that the approximations we made earlier were correct.
Preparation of buffers
Let us now see how we make buffers. Suppose you are asked to prepare a buffer of pH 7.4, the pH of human blood. The first thing you need to do is to select appropriate conjugate acid-base pair. The selection is based on the pKa of the acid; an acid can be used to prepare a buffer within a pH range of pKa ± 1. This means that you cannot prepare an acetate buffer of pH 7.4 because acetic acid-acetate conjugate base pair is a good buffer only for the pH range of 3.76 – 5.76 (see figure 3.1). Let us see the dissociation of phosphoric acid:
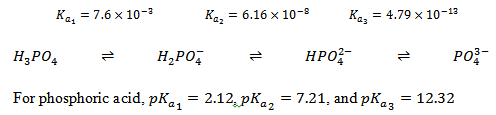 |
(3.19) |
We can therefore see that the pKa2 of phosphoric acid is suitable for preparing the buffer of pH 7.4. Let us see how to prepare a phosphate buffer of pH 7.4.
Example 3.2 : Preparation of 500 ml of 200 mM phosphate buffer of pH 7.4