Methodology: As is evident from equation 3.19, phosphate buffer of
the required
pH can be prepared starting with any of the four chemical species. Suppose we have following four chemicals in the laboratory:
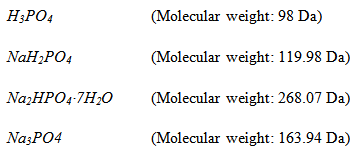 |
We can prepare phosphate buffer of pH 7.4 in the following ways:
- Take the required amount of H3PO4 or NaH2PO4 and dissolve it in 400 ml water. The pH of the solution will be less than 7.4. Titrate the solution with NaOH until a pH of 7.4 is reached; adjust the final volume to 500 ml with water.
- We can take NaH2PO4 and Na2HPO4-7H2O in the amounts that would give a pH of 7.4 when dissolved in water so as to obtain a solution of 500 ml volume. The pH obtained this way, however, is seldom correct.
Okay then. Let's go and prepare it (let us go stepwise):
Calibration of pH meter: pH meters need to be calibrated every time a buffer is to be prepared. A pH meter is calibrated with standard buffers of pH 4.0, 7.0, and 9.2. The pH meter is calibrated with pH 7.0 buffer; the second standard buffer to be used is that of pH 4.0 if the buffer that needs to be prepared is acidic and that of pH 9.2 if the buffer to be prepared is basic.
- Switch “ON” the pH meter and allow it to stabilize for 20 min.
- Take out your standard buffers and allow them to come to room temperature (standard buffers are usually stored at 4°C).
- The electrode of the pH meter is kept in 3 M HCl.
- Take out the electrode and wash it thoroughly with distilled water.
- Measure the pH of “pH 7.0 standard buffer”, set this as pH 7.0 in the pH meter.
- Take the electrode out of the standard buffer and wash it thoroughly with water.
- Measure the pH of “pH 9.2 standard buffer”, set this as pH 9.2 in the pH meter.
Method 1 : Preparing buffer with H3PO4
- Prepare 100 ml of 1 N NaOH solution
- Take 400 ml distilled water in a 1 litre conical flask.
- Add 11.53 ml of H3PO4 dropwise in the 400 ml distilled water
a. The amount of H3PO4 required
b. Concentrated H3PO4 is 85 % (wt/v) with a density of 1.685 g/ml
c. Therefore, the volume of concentrated H3PO4 required:
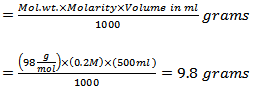
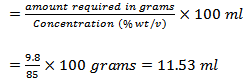
- Wash the pH electrode thoroughly with distilled water and place it in the H3PO4 solution.
- Add 1 N NaOH dropwise; shake well and measure the pH.
- When the pH of the solution nears 7.4, be careful in adding NaOH so that pH does not overshoot.
- Transfer the contents of the flask in a 500 ml volumetric flask.
- Add water to make the final volume of the solution to 500 ml .
- The required buffer is ready.
Note : The same method can be used for preparing the buffer starting with NaH2PO4. The amount of NaH2PO4 required:
![]()
12 g of NaH2PO4 is dissolved in 400 ml water in a 1 liter conical flask and then steps 5-9 are followed.