Titration of a weak acid
Titration of a weak acid (acetic acid) with a strong base (NaOH) is shown in figure 3.1. Addition of NaOH neutralizes the H+ ions present in the solution; thereby increasing its pH. Consumption of H+ ions drives the dissociation of the CH3COOH into CH3COO− and H+ (Le Chatlier's principle). As more and more of NaOH is added, the pH of the solution increases. This increase in pH is small when the solution reaches the pH close to the pKa of acetic acid. A weak acid, therefore, provides a resistance to change in pH near its pKa value. This resistance to change in pH by an acid near its pKa is the concept of a buffer.
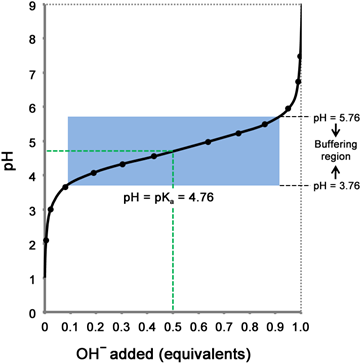
Figure 3.1: Titration curve of acetic acid. Acetic acid is titrated with NaOH and pH is measured after every addition. The pH is plotted against the amount of NaOH expressed as a fraction of the total amount of NaOH required to ionize all the acetic acid. Addition of NaOH drives the dissociation of CH3COOH into CH3COO− and H+. Addition of NaOH causes increase in pH; notice the buffering region (blue) wherein addition of NaOH causes little increase in pH.