Common ion effect
Common ion effect is the change in the ionization equilibrium due to the addition of a solute that provides an ion taking part in the equilibrium. Let us take an example with acetic acid.
Example 3.1 : You are given a 0.1 M solution of acetic acid.
• Calculate the concentrations of all the chemical species present at equilibrium (Given Ka of acetic acid is 1.7 × 10-5M ).
• Calculate the concentrations of all the species when 0.05 M of sodium acetate is added to the solutions.
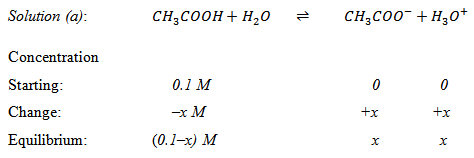
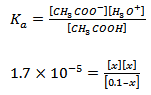 |
(3.9) |
If the acid is <10% ionized i.e. if Ka < 10-3, 0.1–x can be approximate to 0.1. Equation 3.10, therefore, reduces to:
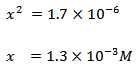 |
(3.11) |
Therefore, the equilibrium concentrations of CH3COOH, CH3COO−, and H3O+ are 98.7 mM , 1.3 mM , and 1.3 mM , respectively.
Solution (b) : Sodium acetate dissociates completely in aqueous solutions; 0.05 M sodium acetate therefore provides 0.05 M of acetate ions.
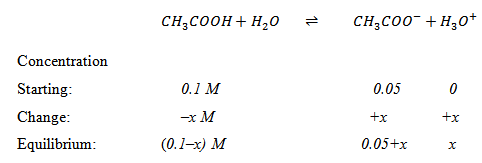
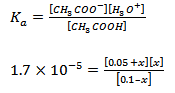 |
(3.13) |