In this lecture, we shall be discussing about the ionization of weak acids and preparation of buffers using weak acids and bases. By the end of this lecture, you should be able to prepare a buffer of any strength (concentration) and any pH.
Ionization of weak acids and weak bases
In biochemistry, it is convenient to use the Brønsted concept of acid and base; a substance that donates protons is an acid while a substance that accepts the protons is a base. When an acid loses a proton, a base is produced. The acid and the corresponding base are said to constitute a conjugate acid-base pair.
| (3.1) |
In the above ionization reaction, HA and A - make one conjugate acid-base pair while B - and HB make another conjugate acid-base pair.
Strong acids like HCl, H2SO4, and HNO3 and strong bases like NaOH and KOH completely dissociate in dilute aqueous solutions. The weak acids and bases are those that do not dissociate completely when dissolved in water. The dissociation of a weak acid in water can be written as a reversible reaction:
| (3.2) |
The ionization can be represented by the equilibrium constant, Keq:
| (3.3) |
Again, the concentration of the undissociated water can be assumed to be constant (55.5 M ). Equation 3.3 can therefore be written as
| (3.4) |
[55.5 M ] Keq is another constant; represented as Ka for the dissociation of acids. The equation 3.4 can therefore be rewritten as:
| (3.5) |
Taking logarithm of equation 3.5
| (3.6) |
Rearranging equation 3.6:
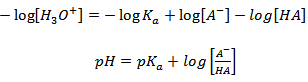 |
(3.7) (3.8) |
where, p is a shorthand for negative logarithm (–log). The equation 3.8 is also known as the Henderson-Hasselbalch equation. From Henderson-Hasselbalch equation, pKa can be defined as the pH at which an acid is 50% ionized.