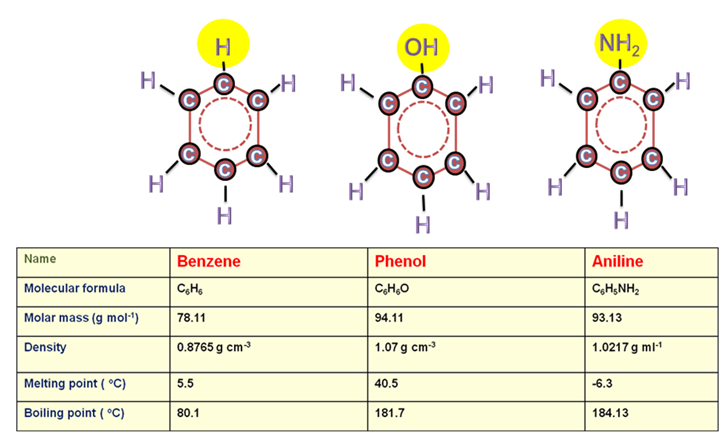
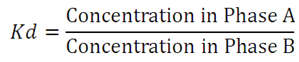 Figure 28.1: Chemical Structure and physical Properties of benzene, phenol and aniline.
Figure 28.1: Chemical Structure and physical Properties of benzene, phenol and aniline.
Principle of Seperation: How a physical or chemical property will allow to isolate a particular substance? The mixture of compound 1 and 3 is shown in Figure 28.2 and assume if we are using boiling point as a criteria to isolate them. As we will heat the mixture there will two phase forms, one liquid phase and other is vapor phase. The molecules of compound 1 and 3 will distribute between these two phases and as the temp is near to boiling point of compound 1, more amount of 1 will be present in vapor phase than liquid phase. Where as more number of compound 3 will be in liquid phase. Eventually as this process will continue, at the end two molecules will get separated from each other. The distribution coefficient (Kd) to describe the distribution of compound 1 between two phase A and B is as follows: